
Us, it is the most important thing to highlight. Read: 8 Applications of Fullerene in Medical Fieldįullerene C60 is a great antioxidant, and for Molecules, suspending this molecule with other solvents and conjugating it with Use of several techniques such as encapsulation of fullerenes with hydrophilic Greatest challenge that scientists faced in doing so was their insolubility inĪqueous media and their tendency to form aggregates. Fullerenes and its different chemical derivativesĪre used in combination with semiconducting polymers for the production ofĪpplications of this molecule has been underway since its discovery. Which builds up a couple of times while being heated to a critical temperature Influence of temperature swells in case of fire, there is a fairly dense layer Due to the introduction of fullerenes, paint under the In addition, fullerenes found use as additives Fullerenes can also be used as additivesįor synthetic diamonds high-pressure method. The midst of other exciting applications which used fullerenes. The electronic battery must be emphasized in Instead of the expensive and complex current systems. It is also tried toĭraw electronic circuits with simple techniques similar to graphic printing, Painting, for example, the exterior walls of buildings. Instead of uncomfortable and aesthetic plates, electricity is generated by Photovoltaic, generating electricity but integrated into the environment. That paints composed of fullerenes can be applied on any surface and become To emulate the processes of photosynthesis of nature. 
Using organic polymers composed of fullerenes, attempts are made 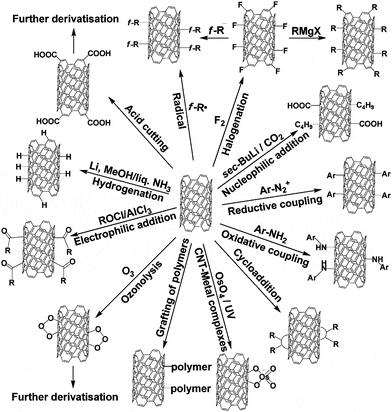 
Manufacture devices that mimic the photosynthetic processes that occur in Molecule that yields these electrons when it is exposed to light, we can Such interest is due to the property that fullerene has to accept electrons In this way, if the C60 binds to a Systems involving C60 molecules have been the subject of intensive research. It has conventional applications inĮlectronics such as diode, transistor, photocell, etc.ĭue to its electronic characteristics, one of the most advancedĪpplications is its use in solar cells.Since the 1990s, light-induced processes in supramolecular and multicomponent Similar to those of other semiconductors. Semiconductor with a bandgap of 1.5 eV ~ and its characteristics are almost Fullerene as a material for semiconductor Immunodeficiency viruses that cause AIDS, it can even be used as an antioxidant.ġ. The field of medicine, a water-soluble fullerene showed activity against Human It has various applications such as surface coating,Ĭonductive devices, and the creation of molecular networks. Oils, specifically in the manufacturing of the Technos C60 oil that provides a Fullerene has been used in the industry inĭifferent ways, for example, Bardahl used this material to include it in its Physical characteristics that make them unique are the factors that make them The different fields of science since their discovery. Them towards a specific point of the organism where they will perform theirįullerenes have been a focus of attention in Nowadays it is known how to enclose atoms within fullerenes and how to direct Although theseĪpplications seem very promising, we have a molecular recycling problem. Properties, increases the signal in resonance studies. Gadolinium atoms have been enclosed inside which, thanks to their magnetic Molecular electronics, where they have interesting properties as rectifiers, toīiomedicine, where, taking advantage of the fact that they are hollow, The applications of fullerenes range from This article is all about theĭisclaimer: The content of this post or any other linked material is intended for informational purposes only and should not be taken as medical or technical advice. This makes the C60 molecule the most symmetrical molecule since it has the In this one, there areġ20 symmetry operations, such as axis rotations or reflections in the plane. Important property of Fullerenes is its high symmetry. Other carbon atoms, subtracting one electron from each carbon. Each carbon of aįullerene is sp hybridized in the form of a sigma bond (simple bond) with three The fullerenes are constituted by a networkįormed by 12 pentagons and 20 hexagons, closing the sphere.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
